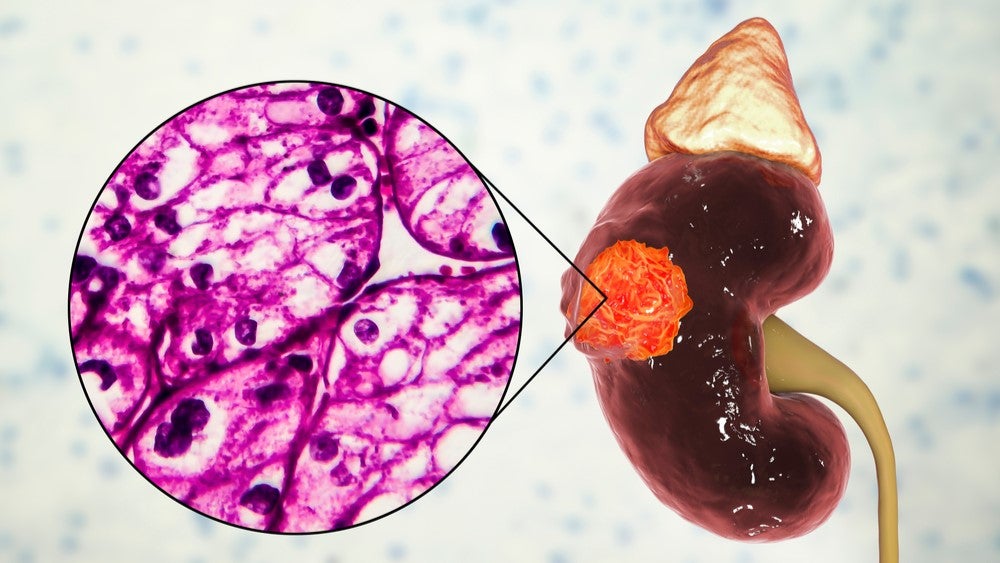
Merck & Co (MSD) and HiberCell have partnered to conduct a Phase Ib trial to evaluate the combination therapy of MSD’s Welireg (belzutifan) and HiberCell’s HC-7366 for treating clear cell renal cell carcinoma (RCC).
Welireg is an oral hypoxia-inducible factor-2α (HIF-2α) inhibitor. It was approved as a treatment for certain patients with von Hippel-Lindau (VHL) disease including those with associated RCC. The drug has received a priority review as a treatment for advanced RCC by the US Food and Drug Administration (FDA), with the Prescription Drug User Fee Act (PDUFA) set for 17 January 2024.
HC-7366 is a selective general control nonderepressible 2 (GCN2) kinase activator. GCN2 kinase is part of an integrated stress response, and its prolonged activation can have an anti-tumour and immunomodulatory effect.
Phase Ib trial design
The Phase Ib study is designed to evaluate the safety, tolerability, and preliminary efficacy of the combination therapy. The trial will have dose-escalation and dose-expansion phases, which will determine the recommended Phase II dose.
Additionally, the study will have an independent HC-7366 monotherapy cohort. The trial will enrol up to 80 participants across multiple US sites. The study is planned to start in Q1 2024.
HC-7366 is also being investigated as monotherapy in Phase Ia/Ib trial (NCT05121948) in advanced solid tumours. The open-label study is expected to conclude in October 2024, as per ClinicalTrials.gov.
HiberCell has previously collaborated with MSD to evaluate the combination therapy of MSD’s Keytruda (pembrolizumab) and HiberCell’s odetiglucan. The combination therapy was evaluated in an open-label Phase II IMPRIME 1 trial (NCT05159778) in patients with heavily pre-treated metastatic breast cancer.
Patients who received the combination therapy had an overall response rate of 15.9% and a median overall survival of 16.4 months, as per a 2022 American Society of Clinical Oncology abstract.
MSD has invested in expanding its portfolio of renal cancer therapies. In October, the company licensed three of Daiichi Sankyo’s antibody-drug conjugate (ADC) candidates. One of the three ADCs included in the agreement was ifinatamab deruxtecan, which is in an open-label Phase II clinical trial (NCT05280470) in patients with pre-treated extensive-stage small cell lung cancer (SCLC).