Ixinity (coagulation factor IX (recombinant)) is an intravenous injection containing recombinant human coagulation factor IX that is indicated for the control and prevention of bleeding episodes in patients suffering from haemophilia B.
The drug was discovered by Emergent BioSolutions and the exclusive rights to manufacture the drug are with CMC Biologics, which will produce the drug at its manufacturing facility in Bothell, Washington.
Ixinity was approved by US Food and Drug Administration (FDA) as an intravenous recombinant human coagulation factor IX therapy for the treatment and management of bleeding episodes in adults and children over the age of 12, affected with haemophilia B, in April 2015.
Haemophilia B causes and symptoms
Haemophilia B is an inherited bleeding disorder that prevents clotting. It is caused due to the absence of coagulation factor IX, which is a clotting protein in the blood.
The disease is characterised by trauma-induced bleeding into joints, muscles or internal organs, either as a result of accidental or surgical trauma. Affected people bleed longer than other people.
Haemophilia B is estimated to affect one in 25,000 male births and there are 4,000 people affected by the disease in the US.
Ixinity’s mechanism of action
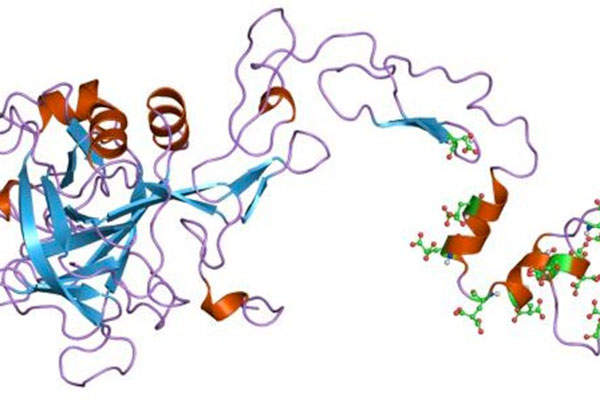
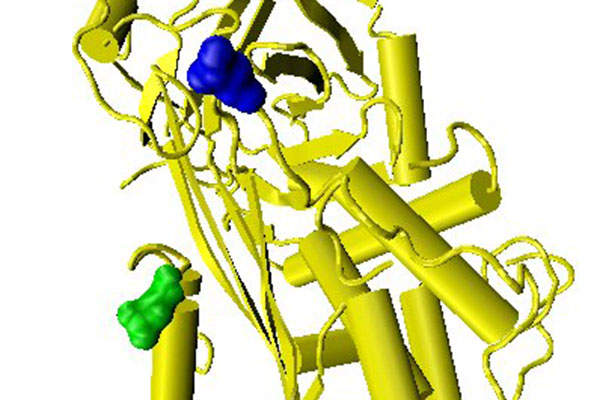
Ixinity is a coagulation factor IX (recombinant), which replaces deficient factor IX and corrects the protein deficiency, reducing bleeding tendency. When administrated, Ixinity increases plasma levels of factor IX and temporarily corrects the coagulation defect in patients.
The drug is available as a lyophilised white powder packed in single-use glass vials containing nominally 500 international units (IU), 1,000IU, or 1,500IU per vial. The powder has to be reconstituted with sterile water for injection through intravenous administration.
Clinical trials on Ixinity
Biogen idec received US Food and Drug Administration’s (FDA) approval for Alprolix in March 2014 for the control and prevention of bleeding episodes, perioperative (surgical) management and routine prophylaxis in adults and children with haemophilia B.
The FDA’s approval of Ixinity is based on results from Phase I/III open-label, uncontrolled, multi-centre, global study conducted to evaluate the safety and efficacy of the drug.
The study enrolled 77 patients who either had severe or moderately severe haemophilia B or had factor IX levels between 2IU/dL – 8IU/dL and clinically severe haemophilia B with recurrent bleeding episodes and required surgery.
Out of 77 patients, 68 were previously treated patients (PTPs) with a minimum of 150 exposures to another factor IX preparation.
Subjects were treated with Ixinity either as routine or on-demand treatment of bleeding episodes. Out of the 68 patients, 55 received Ixinity for more than 50 exposure days and 45 received Ixinity for more than 100 exposure days.
Routine treatment was typically a starting dose of 40IU/kg – 70IU/kg twice a week, whereas doses in the on-demand therapy were 49.9IU/kg – 71.8IU/kg for bleeding episodes.
The mean number of exposure days was 138.2 and median duration on study was 16.2 months for the routine treatment regimen and 14.1 months for the on-demand treatment regimen.
A total of 508 bleeding episodes were treated with Ixinity during the studies, of which 84% were resolved by one or two infusions. Subjects showed a mean incremental recovery of 0.98IU/dL and a mean terminal half-life of 24 hours.
In 84% of cases, reduction of bleeding episodes was rated as excellent or good by subjects. Excellent was defined as a dramatic response with abrupt pain relief and clear reduction in joint or haemorrhage site size, while good was defined as pain relief or reduction in haemorrhage site size that may have required an additional infusion for resolution.
The most common adverse reaction reported in clinical trials was headache. Other adverse reactions included weakness, lack of feeling, depression, haemophilia, influenza (flu), injection site discomfort, lethargy and skin rash.